
Personal Chair
- About
-
- Email Address
- h.deng@abdn.ac.uk
- Telephone Number
- +44 (0)1224 272953
- Office Address
- School/Department
- School of Natural and Computing Sciences
Biography
08/2022 - Personal Chair, University of Aberdeen, UK
08/2018 - 07/2022 Reader, University of Aberdeen, UK
08/2014 - 07/2018 Senior Lecturer, University of Aberdeen, UK
10/2008 - 07/2014 Lecturer, University of Aberdeen, UK
06/2002 - 09/2008 Postdoctoral fellow, University of St Andrews, UK
1999 - 2002 Ph.D. University of Wales Swansea, UK
Memberships and Affiliations
- Internal Memberships
-
Director of Research
PGR coordinator
CM2514 course coordinator
- External Memberships
-
BBSRC pool of expert
MRC and BBSRC panel members
UKRI FLF peer review colleague
External reviewer for a series of peer-reviewed journals within my area of interest, including Nature, Nat. Chen. ACIE, Sci. Adv. Chem. Sci., Chem Comm., ACS Chem. Biol., ChemBioChem, Marine Biotechnology, Org & Biomol. Chem, FEMS Microbiology Ecology, Chemistry today, Marine Drugs, Fish and Shellfish Immunology, and Virulence.
A reviewer of research councils, BBSRC (UK) and FCT (Fundação para a Ciência e a Tecnologia in the field of Chemistry and Biochemistry, 2010-2011), Fonds de recherche du Québec - Nature et technologies, Québec, Canada (2015-2016) and charities, Leverhulme Trust (UK).
- Research
-
Research Overview
My research carries on the great tradition of UK natural product biosynthesis activity. We have all the required skills to succeed in such an enterprise, involving discovery of new families of specialised metabolites, bacterial genomics, chemical synthesis and the associated molecular biology to manipulate genomes to enzymology and the reconstruction of biochemistry. All of this is focussed on understanding the molecular basis and mechanism of natural products assembly.
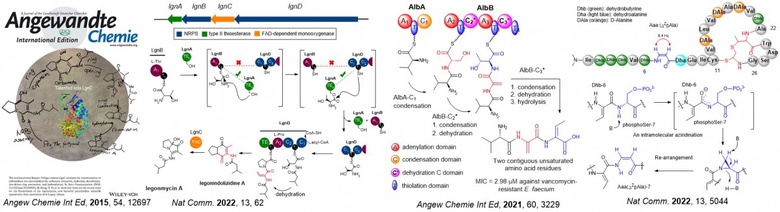
Recent contributions involve the understanding of a group of unsaturated amino acids called dehydroamino acids (dhAAs). dhAAs are key components in many peptidyl therapeutics and versatile building blocks in peptide chemistry where organic chemists generate peptidyl derivatives through Michael additions, cross couplings and cycloaddition. They are used as bio-orthogonal handles for later stage modification of biomolecules. However, accessing dhAAs in structurally complex molecules presents a synthetic challenge, often resulting in poor atomic economy.
Among approximately 40 dhAAs found in the natural product inventory, only two, dehydroalanine (Dha) and (Z)-dehydrobutyrine (ZDhb), are well-studied. In this context our lab has elucidated the formation of several understudied dhAAs in newly discovered bacterial peptide-related metabolites, such as pyrrolizidine alkaloids (PAs) (Angew Chem. Int. Ed. 2015, 54, 12697, Nat. Comm. 2022, 13, 62), short dehydrated non-ribosomal peptides (Angew Chemie. Int. Ed. 2021, 60, 3229) and a new family of ribosomally synthesized and posttranslationally modified peptides (RiPPs) (Nat. Comm. 2022, 13, 5044) as shown below. Apart from this, we have also accumulated an outstanding body of work from bioactive natural product discovery to new enzymology as evidenced in our publication profile.
The discovery we made offers an alternative to rational engineering of pathways to generate bioactive peptides. As such we have obtained grants from various funding bodies (i.e. UKRI, Leverhulm Trust, Royal Society, Royal Society of Edinburgh, IBIOIC, EC) to further investigate potential applications of novel enzymes identified in these pathways towards the development of pharmaceuticals.
Research Areas

Biological and Environmental Sciences

Chemistry
Research Specialisms
- Industrial Biotechnology
- Biochemistry
- Organic Chemistry
- Applied Chemistry
Our research specialisms are based on the Higher Education Classification of Subjects (HECoS) which is HESA open data, published under the Creative Commons Attribution 4.0 International licence.
Past Research
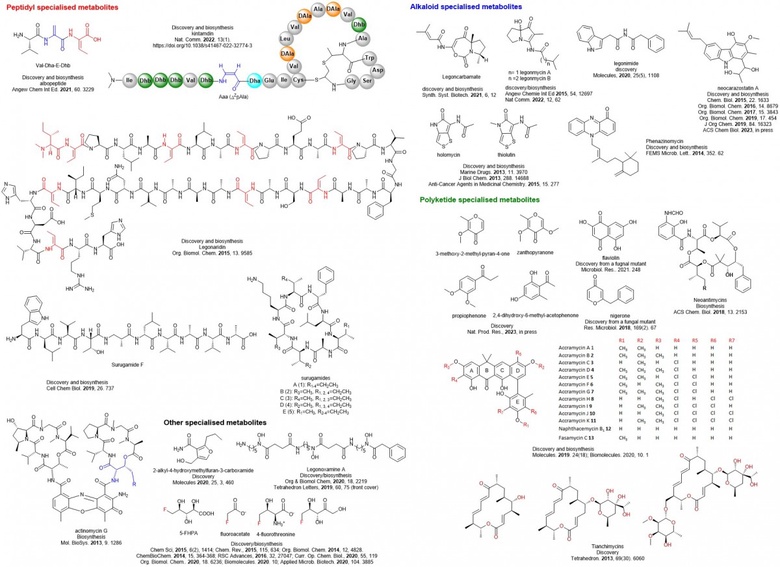
My team has accumulated a good body of work on bioactive natural product discovery (from polyketides and peptides to alkaloids) and their biosynthesis, and new enzyme investigation as shown below. Collectively it is a combination of both its range and illustration and these new organic molecules and new enzymes my team has discovered will find applications for the development of pharmaceuticals and agrochemicals or new ways to biosciences.
Collaborations
Professor Mathew Jenner, Department of Chemistry, University of Warwick, UK
Professor Steven Cobb, Department of Chemistry, University of Durham, UK
Dr Jioji Tabudravu, School of Natural Sciences, University of Central Lancashire, UK
Dr David Clarke, EastChem, School of Chemistry, University of Edinburgh, Edinburgh, EH9 3FJ, UK
Professor Bruce Milne, Department of Physics, University of Coimbra, Rua Larga, 3004-516, Coimbra, Portugal
Professor Yi Yu, School of Pharmaceutical Sciences, Wuhan University, Wuhan 430071, China
Professor Kwaku Kyeremeh, Department of Chemistry, University of Ghana, Ghana
NCIMB Ltd and Ingenza Ltd
Funding and Grants
We gratefully thank the funding bodies below for financial supports of our research
MRC, BBSRC, Leverhulm Trust, EC, IBIOIC, The Royal Society and The Royal Society of Edinburgh.

- Teaching
-
Teaching Responsibilities
CM4518 Biological origin of natural products
CM1020 Chemistry for Biosciences 1
CM1512 Chemistry for Biosciences 2
CM3534 Organic and Biological Chemistry
- Publications
-
Page 2 of 5 Results 26 to 50 of 104
Upconversion NIR-II fluorophores for mitochondria-targeted cancer imaging and photothermal therapy
Nature Communications, vol. 11, 6183Contributions to Journals: ArticlesDiscovery of New Antibacterial Accramycins from a Genetic Variant of the Soil Bacterium, Streptomyces sp. MA37
Biomolecules, vol. 10, no. 10, pp. 1-11Contributions to Journals: ArticlesDefluorination of 4-fluorothreonine by threonine deaminase
Organic & Biomolecular Chemistry, vol. 18, no. 32, pp. 6236-6240Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/d0ob01358g
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/14988/1/Wu_etal_OBC_Defluorination_VOR.pdf
- [ONLINE] View publication in Scopus
Identification of 5-Fluoro-5-Deoxy-Ribulose as a Shunt Fluorometabolite in Streptomyces sp. MA37
Biomolecules, vol. 10, no. 7, 1023Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.3390/biom10071023
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/14704/1/biomolecules_10_01023_v2.pdf
- [ONLINE] View publication in Scopus
Novel South African Rare Actinomycete Kribbellaspeibonae Strain SK5: A Prolific Producer of Hydroxamate Siderophores Including New Dehydroxylated Congeners
Molecules, vol. 25, no. 13, 2979Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.3390/molecules25132979
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/14708/1/molecules_25_02979_v2_1.pdf
- [ONLINE] View publication in Scopus
An unusual metal-bound 4-fluorothreonine transaldolase from Streptomyces sp. MA37 catalyses promiscuous transaldol reactions
Applied Microbiology and Biotechnology, vol. 104, pp. 3885-3896Contributions to Journals: ArticlesFluorine biocatalysis
Current Opinion in Chemical Biology, vol. 55, pp. 119-126Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/j.cbpa.2020.01.004
- [ONLINE] View publication in Scopus
Characterization of the promiscuous N-acyl CoA transferase, LgoC, in legonoxamine biosynthesis
Organic & Biomolecular Chemistry, vol. 18, no. 12, pp. 2219-2222Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/d0ob00320d
- [ONLINE] View publication in Scopus
Novel NIR-II organic fluorophores for bioimaging beyond 1550 nm
Chemical Science, vol. 11, no. 10, pp. 2621-2626Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/C9SC06567A
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/13834/2/Li_etal_CS_Novel_NIRII_VOR.pdf
- [ONLINE] View publication in Scopus
Targeted isolation of indole alkaloids from Streptomyces sp. CT37
Molecules, vol. 25, no. 5, 1108Contributions to Journals: ArticlesThe X-factor: enhanced β-oxidation on intracellular triacylglycerols enabling overproduction of polyketide drug-like molecules in microorganisms
Synthetic and Systems Biotechnology, vol. 5, no. 1, pp. 19-20Contributions to Journals: ArticlesSignalling and Bioactive Metabolites from Streptomyces sp. RK44
Molecules, vol. 25, no. 3, 460Contributions to Journals: ArticlesA Co-Culturing Approach Enables Discovery and Biosynthesis of a Bioactive Indole Alkaloid Metabolite
Molecules, vol. 25, no. 2, 256Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.3390/molecules25020256
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/13522/1/Maglangit_molecules_VOR.pdf
- [ONLINE] View publication in Scopus
MS Dereplication for Rapid Discovery of Structurally New or Novel Natural Products
Marine Drugs, vol. 18, no. 40, pp. 56-57Contributions to Journals: Articles- [ONLINE] http://clok.uclan.ac.uk/31403/
- [ONLINE] DOI: https://doi.org/10.3390/md18010040
- [ONLINE] View publication in Scopus
Enzymatic Reconstitution and Biosynthetic Investigation of the Bacterial Carbazole Neocarazostatin A
Journal of Organic Chemistry, vol. 84, no. 24, pp. 16323-16328Contributions to Journals: ArticlesPaenidigyamycin g: 1-acetyl-2,4-dimethyl-3-phenethyl-1H-imidazol-3-ium
MolBank, vol. 2019, no. 4, M1094Contributions to Journals: Comments and Debates- [ONLINE] DOI: https://doi.org/10.3390/M1094
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/13483/1/Molbank_PaenidigyamycinG_VOR.pdf
- [ONLINE] View publication in Scopus
Accramycin A, a New Aromatic Polyketide, from the Soil Bacterium, Streptomyces sp. MA37
Molecules, vol. 24, no. 18, 3384Contributions to Journals: ArticlesDigyaindoleacid A: 2-(1-(4-hydroxyphenyl)-3-oxobut-1-en-2-yloxy)-3-(1H-indol-3-yl)propanoic acid, a novel indole alkaloid
MolBank, vol. 2019, no. 3, M1080Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.3390/M1080
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/22988/1/Kwain_M_DigyaindoleacidA_VOR.pdf
- [ONLINE] View publication in Scopus
Mn-Loaded apolactoferrin dots for in vivo MRI and NIR-II cancer imaging
Journal of Materials Chemistry A, vol. 7, no. 31, pp. 9448-9454Contributions to Journals: Articles- [ONLINE] http://10.1039/c9tc01929d
- [ONLINE] DOI: https://doi.org/10.1039/c9tc01929d
- [ONLINE] View publication in Mendeley
α-D-glucopyranosyl-(1→2)-[6-O-(L-tryptophanyl)-β-D-fructofuranoside]
MolBank, vol. 2019, no. 2, M1066Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.3390/M1066
- [OPEN ACCESS] http://aura.abdn.ac.uk/bitstream/2164/13123/1/Kveremeh_ADGlucoyranosyl_VOR.pdf
- [ONLINE] View publication in Scopus
- [ONLINE] View publication in Mendeley
Investigation of Penicillin Binding Protein (PBP)-like Peptide Cyclase and Hydrolase in Surugamide Non-ribosomal Peptide Biosynthesis
Cell chemical biology, vol. 26, no. 5, pp. 737-744Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/j.chembiol.2019.02.010
- [ONLINE] View publication in Mendeley
- [ONLINE] View publication in Scopus
Novel electrochemical nanoswitch biosensor based on self-assembled pH-sensitive continuous circular DNA
Biosensors and Bioelectronics, vol. 131, pp. 274-279Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/j.bios.2019.02.004
- [ONLINE] View publication in Scopus
A novel near-infrared fluorescent light-up probe for tumor imaging and drug-induced liver injury detection
Chemical Communications, vol. 55, no. 17, pp. 2541-2544Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/c8cc10286d
- [ONLINE] View publication in Mendeley
In vitro reconstitution of the biosynthetic pathway of 3-hydroxypicolinic acid
Organic & Biomolecular Chemistry, vol. 17, no. 3, pp. 454-460Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1039/c8ob02972e
- [ONLINE] View publication in Mendeley
Legonoxamines A-B, two new hydroxamate siderophores from the soil bacterium, Streptomyces sp. MA37
Tetrahedron Letters, vol. 60, no. 1, pp. 75-79Contributions to Journals: Articles- [ONLINE] DOI: https://doi.org/10.1016/j.tetlet.2018.11.063
- [ONLINE] View publication in Scopus
